
After a period of time, liquid pharmaceuticals lose their efficacy and must be destroyed because they are not sufficiently effective. The needless destruction of these meds raises the cost of the product and can result in a reduced supply of the life saving product. Let’s see how this company used DOE to significantly extend the shelf life of one of their liquid oncology drugs.
Faced with Multiple Problems: Increased Demand, Reduced Supply, Increased Cost
As part of the company’s deployment of Six Sigma, one of their Master Black Belts (MBB) analyzed the shelf life of their top selling liquid oncology drug. Because of its life-saving nature, the company decided to build sufficient inventory to exceed current demand for the product so if a patient needed it, the product would be available.
The approved FDA spec for the product was a lower limit of 96% concentration. If the product concentration fell below 96%, the product must be destroyed. After analyzing historical data, the MBB determined the lower limit of concentration was occurring after just a little under 3 months on the shelf. Once ordered, the hospital generally administered it to the patient upon delivery which was within two days. This eliminated a concern the drug was sitting in the hospital pharmacy too long and would reach the degradation point before being used.
The senior leadership understood that one option for reducing the amount being destroyed was to better link the inventory level with demand using a replenishment pull philosophy. While this would save money, it also created an opportunity of product not being available a small percentage of the time. If that happened, the hospital might order a competitor’s product or the patient might experience a severe setback, both of which were untenable options to senior leadership.
The MBB was charged with the task of assembling an improvement team including people from manufacturing, quality control and R&D. The team’s goal was to develop a process for extending the shelf life of the product by maintaining the required concentration for a longer period of time. The MBB suggested the team consider using DOE to analyze the effect of the various factors impacting concentration and shelf life.
MBB and Team Designed and Ran the DOE
The first thing the MBB did was to have the team brainstorm the various factors which could have an impact on the product’s concentration. These are the factors which seemed to make the most sense to the team:
- Storage container – currently stored in glass vials but could also be stored in plastic ones
- Color of the container – currently in clear vials but could be in a colored or tinted one
- Orientation of the container – currently stored vertically but could be stored at an angle or horizontally
- Exposure to light – currently in a warehouse with fluorescent lighting but could he stored in the dark, low light or other type of lighting
- Temperature – currently in a refrigerated area set at 65 degrees Fahrenheit but could be colder or hotter
- Rotation of vials – currently vials were not rotated but they could be
- Vial closure – currently a rubber stopper but could be another material or sealing method
After some discussion, the team decided they would start with a 7 factor, 2 level, fractional factorial, resolution III experiment. The seven factors were as identified and the 2 levels for each factor were as follows:
- Storage container – glass(1) vs polyethylene terephthalate(2) containers
- Color – clear(1) vs amber(2)
- Orientation – vertical(1) vs 45 degrees(2)
- Exposure to light – current lighting(1) vs dark with lights automatically coming on when someone enters the area(2)
- Temperature – 65 degrees F(1) vs 60 degrees F(2)
- Rotation – no rotation(1) vs automatic 180 degree rotation once a week(2)
- Vial closure – rubber(1) vs silicone(2)
The purpose of the screening DOE is to reduce the number of total runs of your experiment by screening out those variables that are not statistically significant. In addition to using a fractional factorial, you can also use a technique called the Plackett Burman Design. This also allows you to screen out variables that don’t have much of an effect on the response.
When you do a full factorial DOE, you test every unique combination of your independent variables and how they impact your response variable. You also get the maximum amount of information about the process by looking at the main effects and all interactions.
By using a screening DOE, you lose some information, specifically regarding the interactions of your variables. That may or may not be important to you depending on the purpose of your experiment. We refer to that as losing resolution.
Here is the DOE format for running the experiment. All runs were randomized.

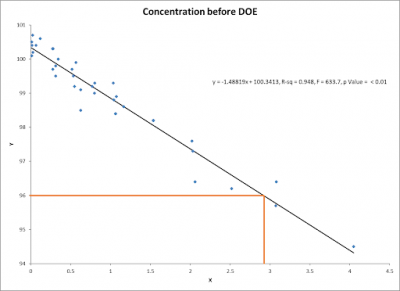
Below is the original scatter plot of the historical data. Note the concentration level is predicted to fall to 96% in a little less than three months. The reduction in concentration seemed to be linear.
Successfully Meeting the Challenge
One of the biggest challenges in running a DOE is to keep out any noise or non experimental conditions from contaminating the results. Fortunately, the company was able to provide a facility where the DOE could be run apart from the normal operations of the company.
Since the actual shelf life had to be simulated, another big challenge was to keep everything running and uncontaminated for the number of months the DOE took. This was accomplished by assigning specific responsibilities to the different team members with the MBB providing frequent oversight as the experiment progressed.
Positive Results and Outcomes Were Beyond Expectations
After analyzing the initial results, the MBB determined that some of the factors were not statistically significant. Of the seven factors, orientation, rotation, closure type and temperature were not statistically significant. The type and color of the vial were important as was the level of light.
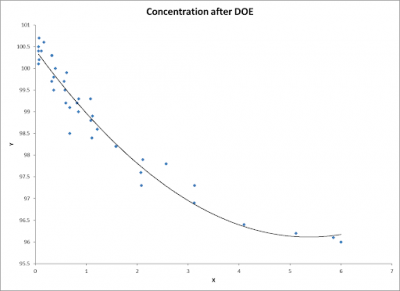
The team decided to repeat the DOE with just the three factors to be sure they had the best prediction and levels. This time they did a 3 factor, 2 level, full factorial experiment with 2 replicates. After analyzing the results of the second experiment, it was concluded that amber glass vials stored in a darkened environment would significantly extend the shelf life of the product. The scatter plot below shows how the rate of concentration reduction was smoothed out and extended the predicted shelf life to approximately six months from the current less than three months.
As a result of this project, the company was able to reduce its cost of product destruction by multi-millions of dollars. This reduction in cost allowed them to reduce their price by 3.8% which made the insurance companies and hospitals very happy.
3 DOE Best Practices
While the company had used DOE in some manufacturing applications, this was the first time they tried it in the context of warehousing and product shelf life. Because of the success of the experiment, the use of DOE in nontraditional applications became more popular. Here are a few concepts to keep in mind when doing a DOE.
1. Use data to identify possible factors and levels
The careful selection of factors is important. If you are using the DMAIC methodology for your continuous improvement effort, your factors might become clear while working in the Analyze phase.
2. Keep out the noise
Don’t let extraneous environmental conditions negatively impact your test results. If your design is sensitive, then any noise will corrupt your results and cause you to draw misleading conclusions about your process or experimental outcomes.
3. Use screening experiments to reduce cost and time
Once you get above 6 or 7 factors, you should consider doing fractional or screening experiments to eliminate non significant factors. This will help reduce the time and expense of your initial experiment. You don’t want to spend a large portion of your budget just to find out only a small number of factors are important. Once you have reduced your factors by using a fractional screening then you can redo the experiment as a full factorial using just the significant factors you found from your screening experiment.
Saving Money and Lives With Basic Six Sigma Statistical Tools
The company was faced with the expensive destruction of product because of limited shelf life. Over time, the process of destroying product became an accepted business practice. As a result of deploying Six Sigma in the company, people’s eyes were open to questioning and challenging how the company handles its business challenges.
Rather than just rely on intuition and gut feelings, the company’s MBB encouraged the company leadership to start using statistical thinking to make decisions. Given the high visibility of the costly destruction of their premier liquid oncology product, the company’s leadership felt it was a good candidate for improvement.
While the use of DOE is more complex than other statistical tools, the MBB and improvement team designed and executed a series of experiments which resulted in a significant increase in shelf life. The result was a savings of millions of dollars in destruction costs and at the same time enabled the company to lower their price and have adequate inventory for hospitals to use for their cancer patients.